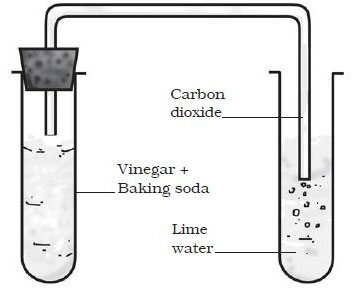
Multimedia: Controlling the Amount of Products in a Chemical Reaction | Chapter 6, Lesson 2 | Middle School Chemistry

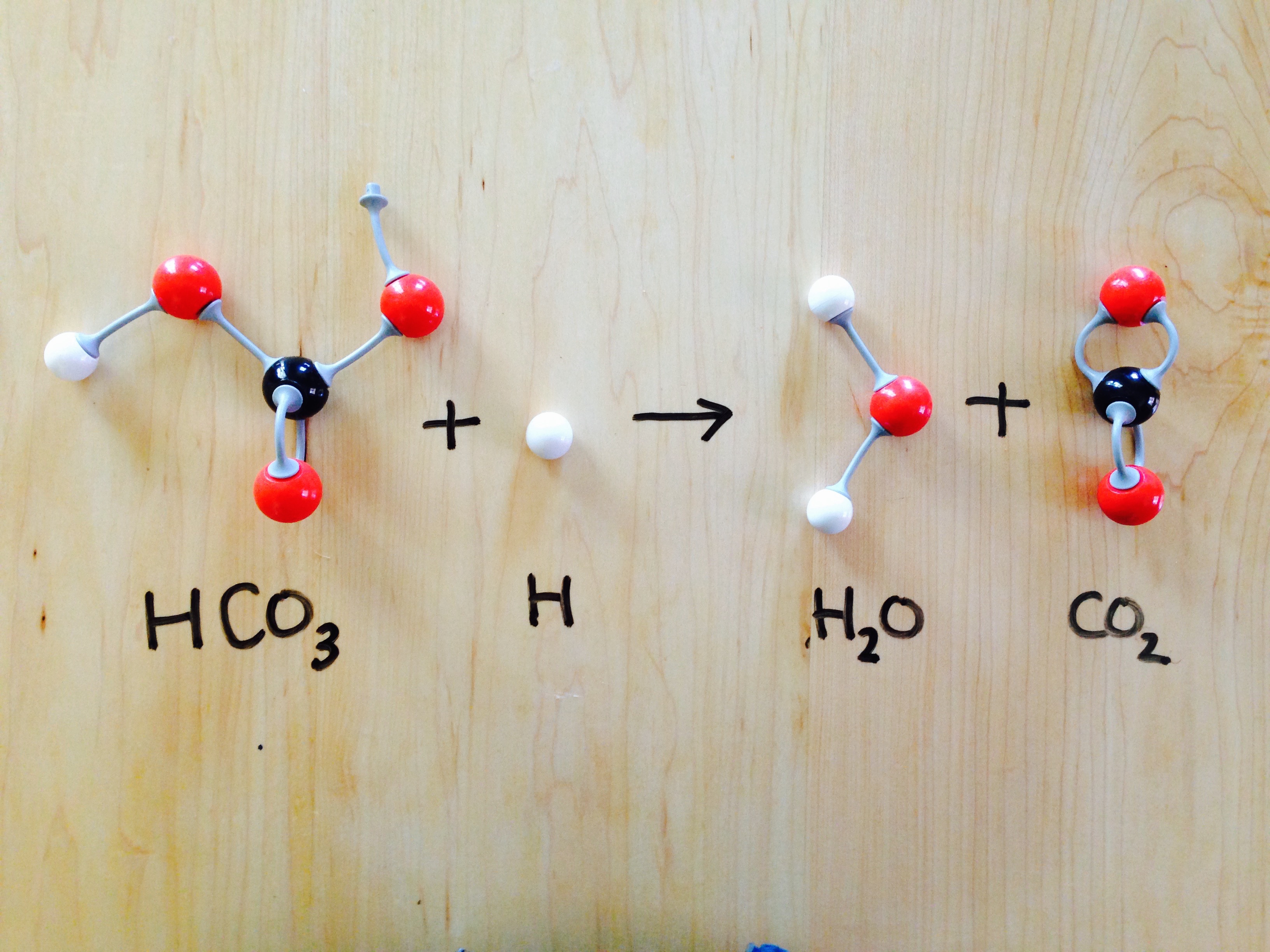
SOLVED: 1.The chemical equation for the reaction of baking soda (sodium bicarbonate, NaHCOs) and vinegar (acetic acid; CH;COOH) may be written as two steps: Fill in the missing information for the chemical



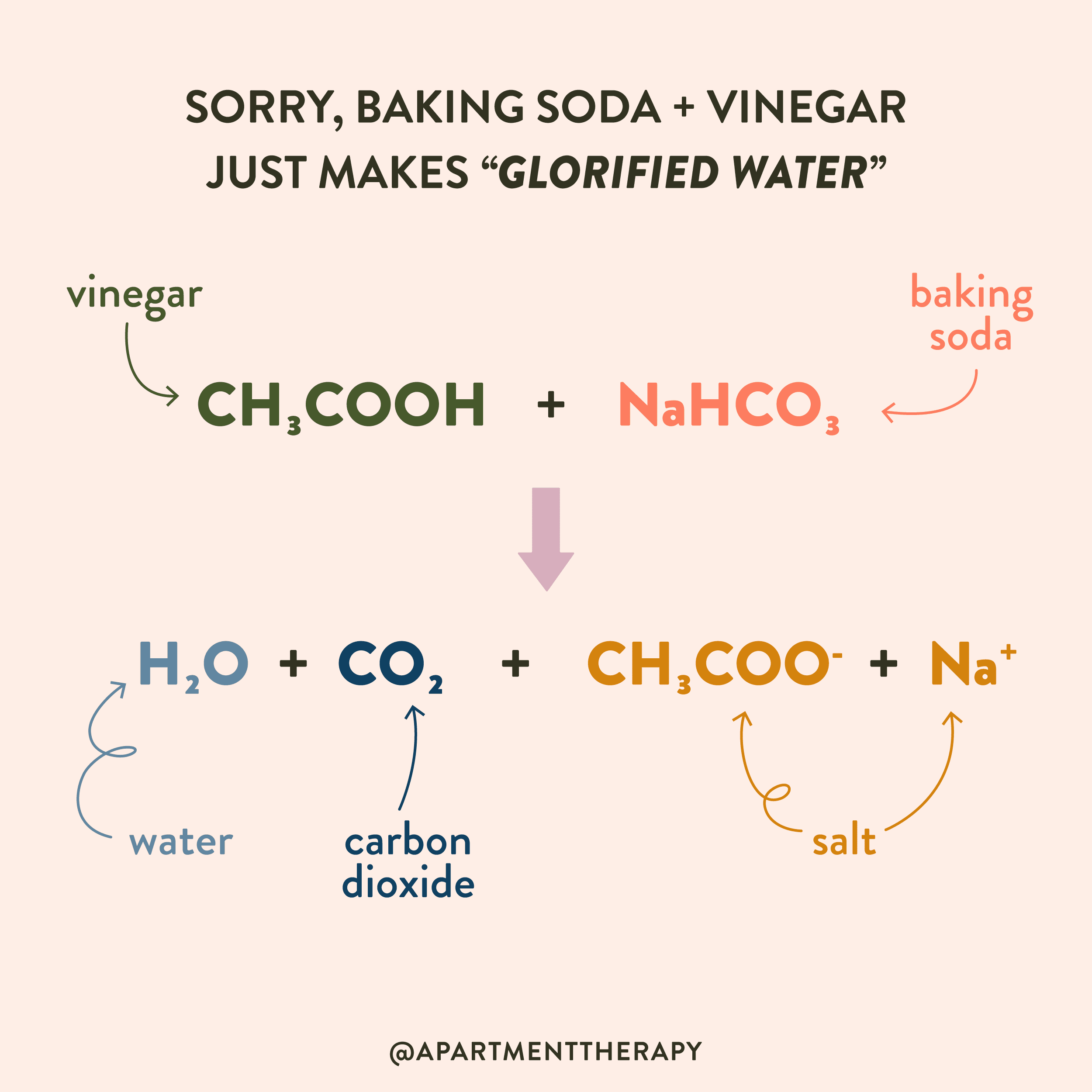



:max_bytes(150000):strip_icc()/Acetate-anion-3D-6dfa0f748a6c47ed93c2aa1b2a64e47e.jpg)


:max_bytes(150000):strip_icc()/sodiumbicarbonate2-599f0a4cb501e800113dd78f.png)